Monoclonal Antibody development
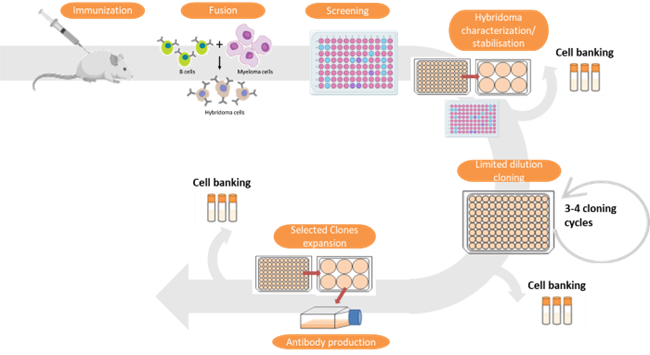
To develop customized monoclonal antibodies, we adapt our protocols at each critical stage of antibody production. This includes designing the antigen, immunization protocol, and screening design of hybridoma cell supernatants. The initial step involves immunizing mice or rats with a specific antigen. We can develop monoclonal antibodies against various antigen types, such as proteins, peptides, small molecules, or cells. The immunization process typically spans 10-12 weeks. The animal with the most robust immune response undergoes a final immunization to stimulate antibody-producing cells. Spleen cells are isolated and fused with immortal myeloma cell lines to create hybridoma cells. The chosen hybridoma clones are then subcloned to ensure cell stability and maintain their monoclonal characteristics. Finally, the isolated cells are expanded and cryopreserved.
Customer benefits
- Versatility: Monoclonal antibodies can be used in diagnostics, therapeutics, and research.
- Customization: Protocols can be adapted to meet specific needs
- Quality Assurance: Our processes adhere to the ISO 9001 quality requirements.
Target customer
- Organizations involved in research and development, pharmaceutical companies, academic institutions, and diagnostic laboratories benefit from monoclonal antibody development.
- Researchers studying diseases, drug targets, and immune responses rely on mAbs for their work.
Additional information
Selected references:
- Giovanna Roncador; Pablo Engel; Lorena Maestre; et al; Alison H.Banham., Nuria Pascual 2016. The European antibody network’s practical guide to finding and validating suitable antibodies for research. mAbs. Taylor & Francis Online. 8-1, pp.27-36
- Rodriguez-Urretavizcaya, N. Pascual, C. Pastells, M. T. Martin-Gomez, Ll. Vilaplana, M.-P. Marco. Diagnostic and Stratification of Pseudomonas aeruginosa Infected Patients by Immunochemical Quantitative Determination of Pyocyanin from Clinical Bacterial Isolates. Frontiers in Cell. Infect. Microbiol., 11, 786929, 2021. DOI: 10.3389/fcimb.2021.786929








