Combination of stem cell therapy and laparoscopy for the treatment of congenital hernia
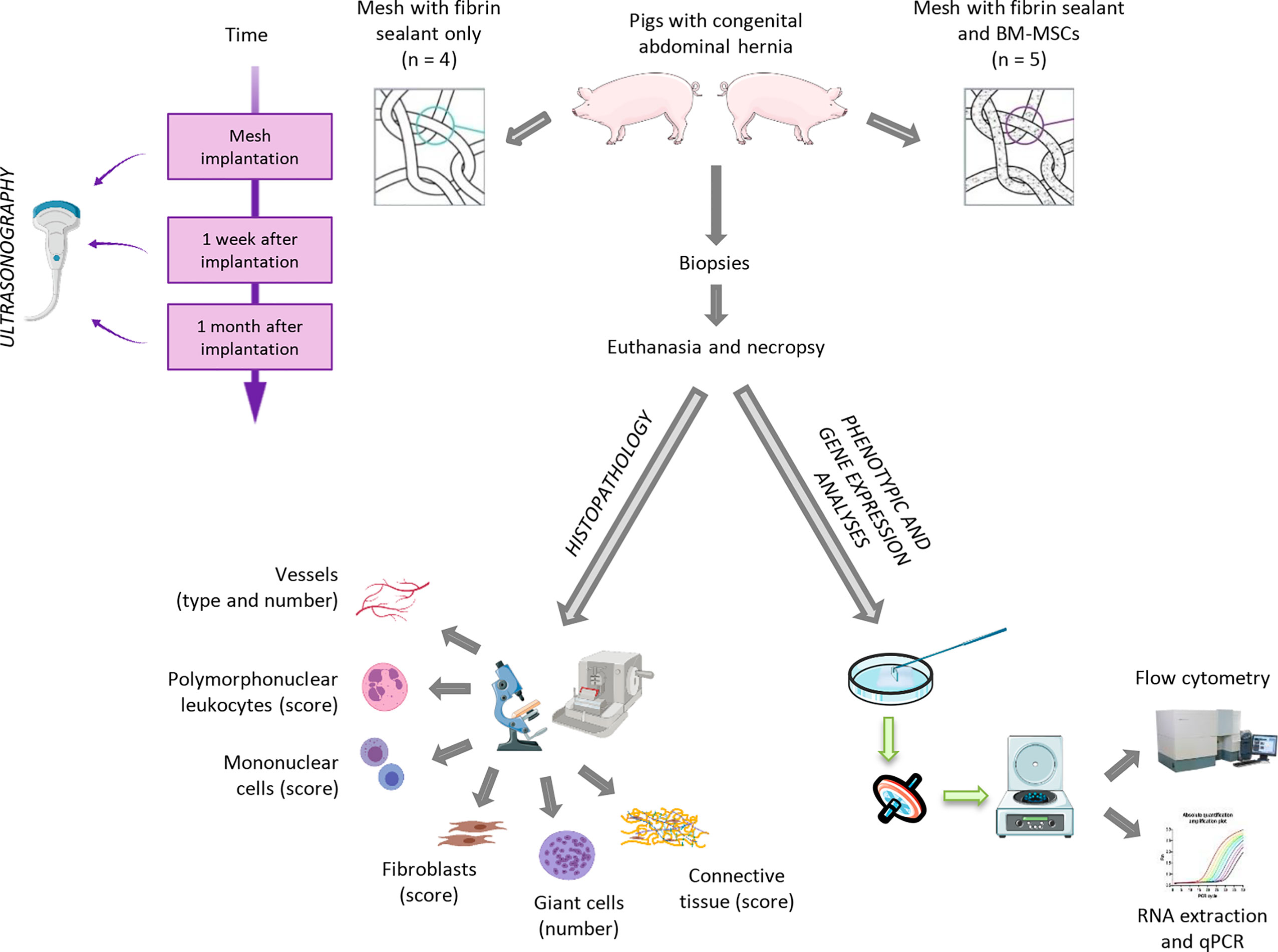
Researchers of CCMIJU and NANBIOSIS Unit 14 Cell Therapy, led by Javier García Casado, have recently published a study with the purpose to combine stem cell therapy and laparoscopy for the treatment of congenital hernia in a swine animal model proposing an easy and feasible method to combine stem cell therapy and minimally invasive surgical techniques for hernia repair, the first preclinical study evaluating the use of stem cell therapy in the field of abdominal hernias in a clinically relevant swine model with congenital hernia
Surgical procedures and imaging diagnoses were performed at the ICTS NANBIOSIS Unit 21 Experimental operating rooms, led by Francisco M. Sánchez Margallo
“We developed an experimental approach to test whether the use of stem cells for abdominal hernia treatment is viable in a clinically relevant animal model. To the best of our knowledge, this is the first preclinical study where pigs with congenital abdominal hernias were treated with surgical meshes seeded with adult stem cells. Additionally, the surgical approach was performed with minimally invasive procedures to avoid complications related to open surgery. An exhaustive follow-up was performed at different time points using different evaluation methods: ultrasonography, gene expression analysis, complete histological evaluation, and cellular characterization by flow cytometry of infiltrated leukocytes.”
Article of reference:
Laparoscopy for the Treatment of Congenital Hernia: Use of Surgical Meshes and Mesenchymal Stem Cells in a Clinically Relevant Animal. Federica Marinaro, Javier G. Casado, Rebeca Blázquez, Mauricio Veloso Brun, Ricardo Marcos, Marta Santos, Francisco Javier Duque, Esther López, Verónica Álvarez, Alejandra Usón and Francisco Miguel Sánchez-Margallo. ModelFront. Pharmacol., 25 September 2020 https://doi.org/10.3389/fphar.2020.01332