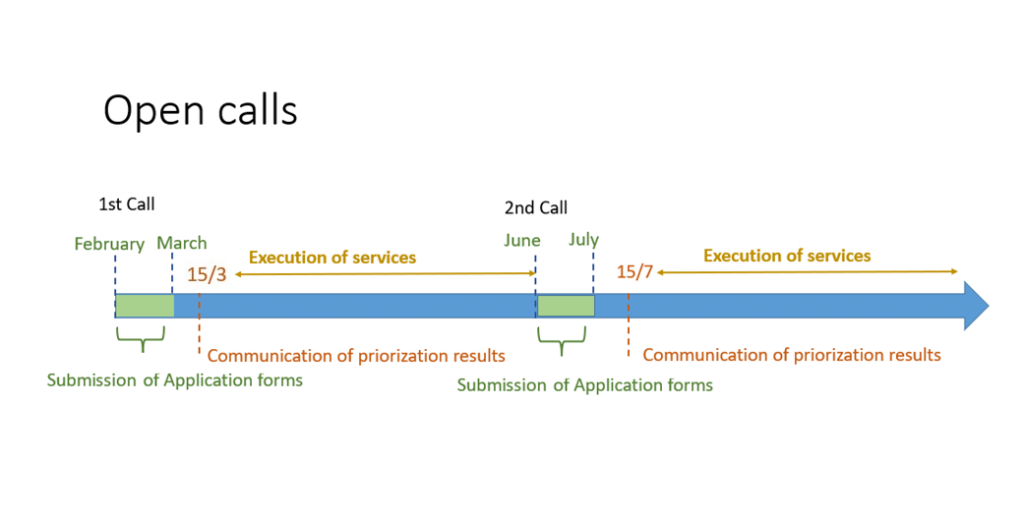
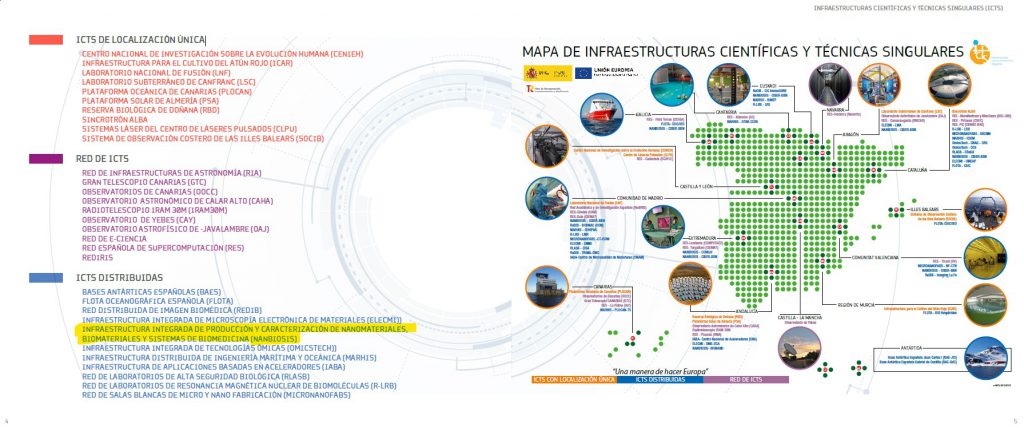
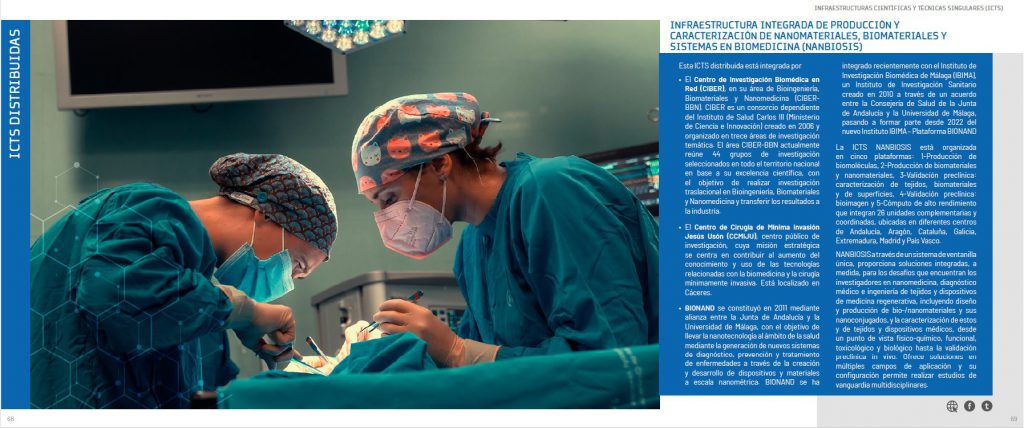
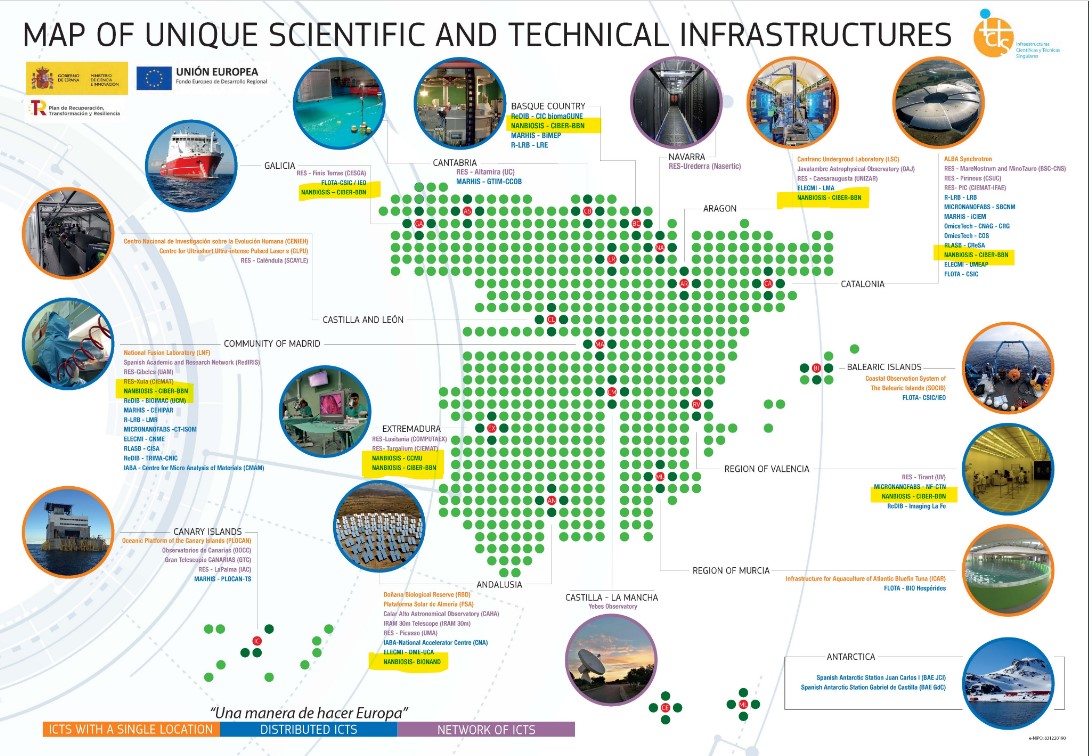
NANBIOSIS receives the support of the Spanish Research Agency (Ministry of Science and Innovation) financing the NANBIO-ACCESS project, in the 2022 call for Excellence Research Networks.
Today has been published the Resolution of the Presidency of the State Research Agency of the Spanish Ministry for Science and Innovation granting fund for the selected projects in the 2022 call for “RESEARCH NETWORKS” within the framework of the State Program to Promote Scientific-Technical Research and its Transfer, of the State Research Plan Scientific, Technical and Innovation
The final resolution proposal was issued on June 21, 2023, once the hearing process has been completed and prior acceptance by the interested parties.
The two years lasting project RED2022-134685-I “NANBIO-ACCESS” was among the selected proposals.
The main goals pursued by the projects are:
1. Promote and consolidate the offer of the NANBIOSIS strategic services, which target advanced challenges in biomedical research: our Cutting-Edge Biomedical Solutions.
2. Promote the open and competitive access to NANBIOSIS services and, especially, to our strategic services.
3. Strengthen NANBIOSIS communication tools and enhance internationalization capabilities.
The proposed activities address the coordination within NANBIOSIS and the complementarity of the capacities of its units, optimizing its resources. Actions are especially proposed to promote the participation of NANBIOSIS nodes and units to the “Cutting-Edge Biomedical Solutions” program.